
The three p orbitals on nitrogen are all mutually perpendicular (or orthogonal) to each other. A p orbital lies along a particular axis: x, y or z. In considering the interaction of two p orbitals, we have to keep in mind that p orbitals are directional. The new orbital produced has a shorter wavelength than the original orbital. There is an antibonding combination in which the orbitals are out of phase.The new orbital produced has a longer wavelength than the original orbital. There is a bonding combination in which the orbitals are in phase.Two original orbitals will combine and rearrange to produce two new orbitals. The combination of one s orbital with another is just like in hydrogen.This is not always true, but the situation varies depending on what atoms we are dealing with. Does the s antibonding combination go higher in energy than the combinations from p orbitals? Do the p bonding combinations go lower in energy than the combinations from s orbitals? We will simplify and assume that the s and p levels remain completely separate from each other. When the orbitals mix, one combination goes up in energy and one goes down. p z orbitals interact with p z orbitals.Īnother complication here is that the s and p orbitals do not start out at the same energy level.p y orbitals interact with p y orbitals.p x orbitals interact with p x orbitals.We can already see how that will work out in dinitrogen, because that is what happened in dihydrogen. For now, we can simplify and say that orbitals on one atom only interact with the same type of orbitals on the other atom. Orbitals interact most easily with other orbitals that have the same element of symmetry. Orbital interactions are governed by symmetry.That process could be extremely complicated, but: 
We need to look at the interaction between the s and p x, p y and p z orbitals on one nitrogen atom with the s and p x, p y and p z orbitals on the other nitrogen. There are three possible atomic orbitals in the 2p level where some of these electrons could be found: p x, p y and p z. Nitrogen has five valence electrons, and these electrons are found in the 2s and 2p levels. A quantitative molecular orbital calculation with a computer would not take this shortcut, but would include all of the electrons in the atoms that are bonding together. In our qualitative examination of bonding in main group diatomics, we will take the approach used in Lewis structures and just look at the valence electrons. 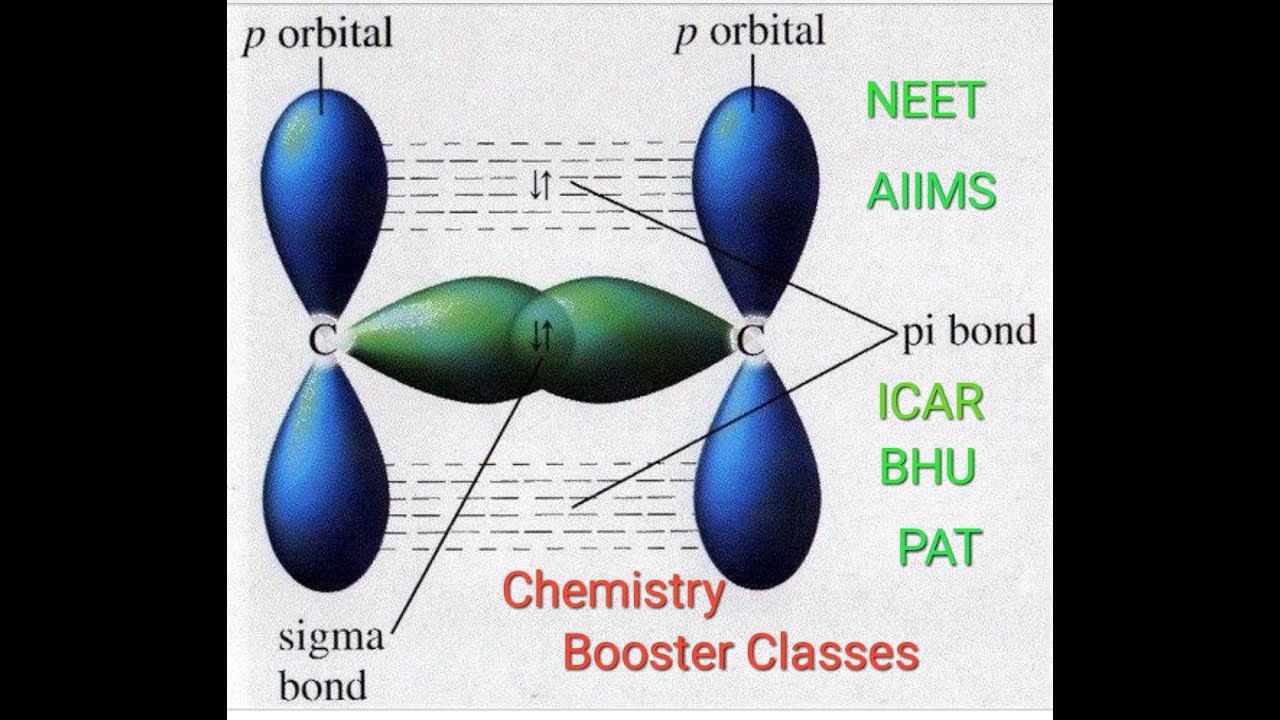
Nitrogen has more electrons than hydrogen, so this interaction is more complicated. We can think about how dinitrogen would form if two nitrogen atoms were placed close enough together to share electrons. Other diatomic molecules in the upper right corner of the periodic table can be constructed in a similar way.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
